Manipulating the gut microbiome: the potential of poo Understand article
This medical treatment might sound repulsive, but don’t pooh-pooh it just yet.

Production / shutterstock.com
/ Nicola Graf
Faeces, stool, poo – whatever you call it, the thought of transferring someone else’s into your own body certainly sounds disgusting. But for someone suffering from a Clostridium difficile infection, a potentially fatal bowel condition, a faecal transplant could save their life. So before you dismiss the idea, consider the reasoning behind this unusual medical treatment: stool contains one crucial component – beneficial bacteria. And the success rate for treating C. difficile infections with a faecal transplant is over 90%. What’s more, scientists at the European Molecular Biology Laboratoryw1 (EMBL) now think that careful matching of donors to patients could make faecal transplants still more effective and widely applicable.
Bacteria and the gut microbiome
When your doctor prescribes antibiotics, you expect the drugs to treat the infection, not to cause a new illness. As well as killing the target bacteria, however, antibiotics (particularly broad-spectrum antibiotics) destroy beneficial bacteria, causing an imbalance in the complex community of micro-organisms in our intestines, known as the gut microbiome.
C. difficile is present in soil, water and air, and lives harmlessly in the guts of roughly one in every 30 healthy adults. But when the normal balance of gut microbes is skewed and there are fewer beneficial bacteria to keep the gut in check, C. difficile can quickly spread.
As it multiplies and grows in the gut, C. difficile produces toxins that cause diarrhoea. When the bacteria are passed out of the body, they can easily infect other people. This makes C. difficile a big problem in hospitals and a major healthcare-associated infection. Other common symptoms include abdominal pain and fever; in severe cases, C. difficile can cause dehydration, inflammation of the intestine, and even a ruptured colon.
For most patients, the infection can be treated with a course of antibiotics that specifically target C. difficile. But in about 20% of cases, the symptoms return, requiring further treatment. Treating recurrent C. difficile is becoming increasingly difficult, as new and resistant strains of the bacterium emerge. One final option for patients fighting the superbug is to undergo a faecal transplant.
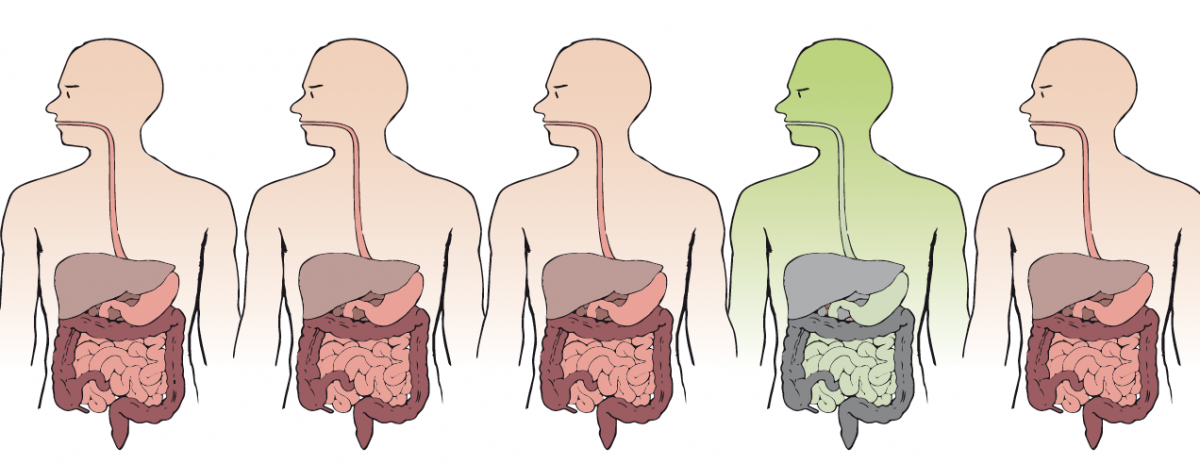
Image courtesy of Nicola Graf
Poo to patient
The screening process for potential stool samples is rigorous: only 3% of volunteers donating samples to the OpenBiome stool bank, for example, are acceptedw2. A stool transplant carries the risk of passing on an infectious disease, and with growing evidence linking the microbiome to obesity, diabetes and allergies, it is possible that these conditions might also be transferred to the patient. In one case, a woman who was successfully treated for a C. difficile infection encountered a surprising side-effect after receiving a stool sample from an overweight donor: she rapidly gained weight herself (Alang & Kelly, 2015). Although the transplant may not have been the only cause, the case raises questions about the role of gut bacteria in metabolism and health.
If a stool sample is deemed suitable, it is liquidised and usually administered via a colonoscopy. The community of micro-organisms from the healthy donor, along with all their genes and metabolic functions, can then begin resetting the balance of the infected patient’s microbiome.
Although the success rate for curing C. difficile infections with faecal transplants is over 90%, the use is still rare – probably due to its unusual nature and our aversion to it. Our faeces, just like blood or vomit, can contain disease-causing organisms, so it’s no surprise that humans want to avoid it, let alone ingest it. Tighter rules on carrying out faecal transplants are also holding back its use, as is the treatment’s invasiveness compared to antibiotics.
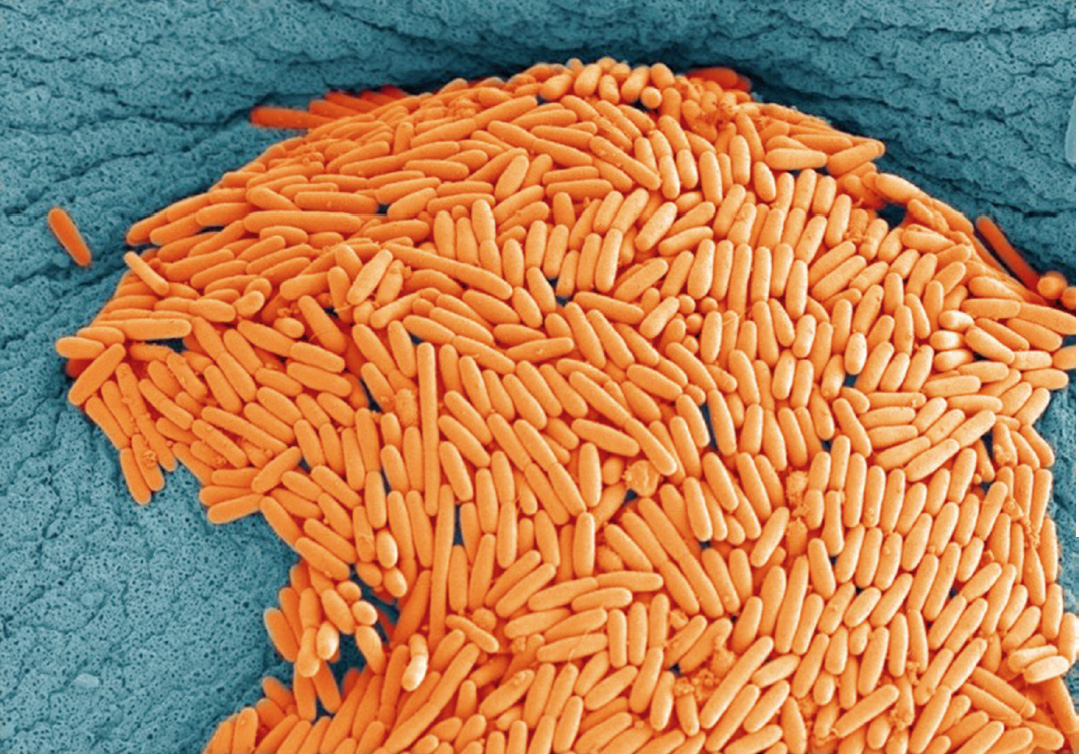
Image courtesy of Wellcome Images (CC BY-NC 4.0)
A personalised pill
To improve their appeal, faecal transplants are moving away from more invasive delivery methods. Instead, patients can swallow something more aesthetically pleasing and manageable: a pill, dubbed a ‘crapsule’. A recent study led by EMBL scientists, with collaborators at Wageningen University and the Academic Medical Centre, both in the Netherlands, and the University of Helsinki, Finland, has also highlighted the need for a tailored approach (Li et al., 2016).
Rather than looking at what species of bacteria inhabit a patient’s gut, the key is to go one step further and see what strains of each species are present. The study found that new strains of bacteria from the donor were more likely to colonise a patient’s gut if the patient already had that species. Simone Li, who carried out the work at EMBL, says the goal is to prescribe a “personalised bacterial cocktail, rather than a one-size-fits-all solution”. Carefully matching donors to patients could improve the effectiveness of faecal transplants.
The demand for faecal transplants doesn’t stop there. Scientists are trying to determine whether transplants could be used to treat other common conditions linked to a skewed microbiome, including allergies, obesity and type 2 diabetes (Bull & Plummer, 2014). And who knows? In the future, we could all be storing healthy poo for later use, and swallowing pills of frozen faecal matter from our personal stool banks.
References
- Alang N, Kelly CR (2015) Weight gain after fecal microbiota transplantation. Open Forum Infectious Diseases 2(1): ofv004. doi: 10.1093/ofid/ofv004
- Bull MJ, Plummer NT (2014) Part 1: The human gut microbiome in health and disease. Integrated Medicine 13(6): 17–22
- Li S et al. (2016) Durable coexistence of donor and recipient strains after fecal microbiota transplantation. Science 352(6285): 586–589. doi: 10.1126/science.aad8852
Web References
- w1 – EMBL is Europe’s leading laboratory for basic research in molecular biology, with its headquarters in Heidelberg, Germany.
- w2 – OpenBiome is a non-profit organisation dedicated to expanding safe access to faecal transplants.
Resources
- Read more about the recent study on improving faecal transplants with a personalised approach on the EMBL news page.
- For more information on faecal transplantation, visit the Johns Hopkins Medicine website.
- To learn more about faecal transplants, read the article ‘Medicine’s dirty secret’ published in Mosaic magazine.
- German scientist and best-selling author Guilia Enders reveals the latest science on our digestive system, including our gut bacteria, in her book Gut. See:
- Enders G (2015) Gut 1st edition. Vancouver, Canada: Greystone Books. ISBN: 978-1771641494
- To understand humans’ dislike towards poo, read the article ‘Why do humans hate poo so much?’ from BBC Future.
Review
Poo is an amusing subject for students to talk about with friends, and the ‘disgusting’ topic of faecal transplants is a good introduction to spark their interest in physiology and learn about different metabolic and physiological processes. Not only is this article relevant to medical sciences, but it could also be used to link to topics such as microbiology and ecology.
Bartolome Piza, CC. Pedro Poveda, Balearic Islands, Spain





