Sugary insights into worm parasite infections Understand article
Schistosomiasis is the second most socioeconomically devastating parasitic disease after malaria. Alan Wilson and Stuart Haslam investigate new ways to combat the parasite – taking advantage of its sugar coating.
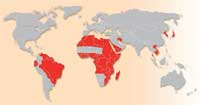
of schistosomiasis. Click to
enlarge image
Image courtesy of Percherie;
image source : Wikimedia
Commons
Schistosomiasis is a major parasitic disease (also known as bilharzia) which infects humans and domestic livestock, and is caused by several species of flatworm in the genus Schistosoma. The World Health Organization estimates that as many as 200 million people are infected in parts of South America, Africa and Asia. Approximately 280 000 people die from schistosomiasis each year in sub-Saharan Africa and millions more are chronically ill. Typical symptoms include abdominal pain, diarrhoea, fever, anaemia and fatigue; the stunting of children’s growth and cognitive development is another consequence of infection. Schistosomiasis thus remains an important public health problem in developing countries.
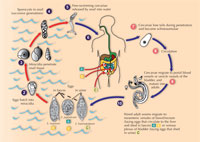
The flatworms pass through a number of stages: (see life cycle) eggs, free-swimming larvae (miracidia), sporocysts, a second free-swimming larval stage (the cercaria) and finally the adult worms. An entire life cycle takes a minimum of 12 weeks.
flatworm parasite
Schistosoma. Click to enlarge
image
Image courtesy of Public
Health Image Library
Worm eggs are released into water when human faeces or urine enter rivers or other water bodies. Freshwater snails of various genera act as intermediate hosts of the flatworms, and the presence of suitable snail species determines the distribution of the disease. Contact with water causes the eggs to hatch in a matter of minutes into miracidia, which enter the snail by penetrating its foot, after which the larvae are known as sporocysts. The sporocysts undergo asexual reproduction within the snail to produce thousands of cercariae, the aquatic stage that infects humans by penetrating the skin. A single snail can shed cercariae for weeks, and this represents a major amplification step in the life cycle of the parasite: one miracidium gives rise to tens of thousands of cercariae before the infection is spent. The most common way of acquiring schistosomiasis is by wading or swimming in lakes or other water bodies that are infested with infected snails. Non-human hosts include other mammals, as well as birds and crocodiles.
Within the human host, the larvae migrate through the blood circulatory system to the hepatic portal blood vessels between the intestine and liver (in the case of Schistosoma mansoni or S. japonicum) or vesical veins of the bladder (in the case of S. haematobium). There, they feed on red blood cells, develop to adulthood and mate. The centimetre-long male then grasps the longer and thinner female in his ventral groove, where she will remain, and, using a combination of oral and ventral suckers, transports her against the blood flow to the smaller blood vessels of the host. Here she pushes forward, poking out at the front of the male’s groove, to deposit hundreds of eggs per day into the blood vessels. Once established, the adult worms can live for decades in the hostile environment of the host bloodstream, potentially open to immune attack. Finally, the eggs must escape into the intestine or the bladder, to be shed to the external environment in faeces or urine, continuing the life cycle.
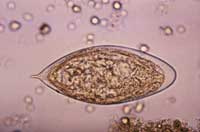
egg, magnified 500x
Image courtesy of Public
Health Image Library
The adult and larval worms are comparatively harmless to their human host, but the eggs can cause severe disease. The severity of the tissue damage caused by the eggs is positively correlated both to the number of worms that a person accumulates, and to the intensity of the human immune response to the eggs: too strong an inflammatory response ultimately leads to more tissue damage, too little to tissue necrosis by egg products. Moreover, a large proportion of the eggs do not escape from the host. Instead, in S. mansoni and S. japonicum infections, they are carried in the blood circulation to lodge in the liver. As a result, fibrous layers of cells from the immune system (known as granulomas) form around the eggs – and it is this response rather than the worms themselves that causes the life-threatening syndrome. S. haematobium eggs are equally dangerous, causing fibrosis – the formation of excess fibrous connective tissue – in the bladder wall.
We were interested in how the parasite enters and leaves the human body. In both the cercarial stage penetrating the host’s skin and the egg escaping through the gut or bladder wall, the parasite releases secretions to help it move through the host’s body. The cercariae possess a series of specialised gland cells. These release a mixture of proteins that have been shown to help the larvae pass through the tough stratum corneum – the outermost layer of the skin – then cross the dermis, and finally penetrate a blood vessel. The secretions from the eggs are released by a specialised tissue, the envelope, which lies beneath the egg shell and completely surrounds the growing miracidium larva inside. These secretions help the eggs to leave a blood vessel and passively cross the tissues to reach the lumen of the intestine or bladder. They are too big to cross capillary beds, so if they break free in the blood vessels they travel downstream to the next organ – the liver, in the case of S. mansoni.
In the long term, the cercarial secretions could be a suitable target for a drug to treat schistosomiasis or for a vaccine to prevent it. But to develop an effective drug, scientists need to know how the secretions work and what they consist of. We characterised the proteins in both the cercarial and egg secretions of S. mansoni using mass spectrometry (see box) and showed that they have a relatively simple composition. Cercarial secretions contain several enzymes that degrade proteins (called proteases), plus a series of proteins and glycoproteins that may function by modifying the host’s immune response. The proteins secreted by the eggs also have protein-degrading activity, although we do not know exactly how they work, because the amino-acid sequence of the principal components is unlike that of any other proteins for which we know the function.
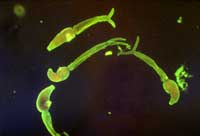
mansoni
Image courtesy of Public
Health Image Library
Unusually for a parasite, both the egg and cercarial secretions are very immunogenic – they provoke strong antibody responses from the host immune system. But what makes the secretions so immunogenic? Studies on human, primate and rodent responses to the infection (Kariuki et al., 2008) have revealed that the vast bulk of antibodies directed against both larval and egg secretions recognise the carbohydrate (glycan) rather than the protein part of the secreted glycoproteins. So what are glycoproteins? And what is the role of the glycans in the biology of the parasites?
The central dogma of modern biology states that DNA encodes the basic template of life, and that the information in the DNA code is first translated into mRNA and finally into proteins which carry out many of the fundamental tasks both in and between the billions of cells which make up a living organism as complex as a human being. But to say that there are just three key types of molecules in living systems is an over-simplification. It is estimated that more than half of all proteins in humans are modified by the addition of sugar molecules, forming glycoproteins. These glycoproteins play a major role in the way that molecules and cells recognise each other, and therefore in the many interactions that determine how diseases are spread or combated.
Every cell in the human body (indeed, in all eukaryotes) is coated with a sugar-rich layer called the glycocalyx. Acting as identity tags, glycans on the outside of the glycocalyx interact with a variety of receptors (recognition molecules) on the membranes of surrounding cells and thereby help to control the social (correct) and anti-social (errant) behaviour of our cells. The worm parasite appears to exploit this glycan recognition process to manipulate the host’s immune system and allow the worm to complete its life cycle. By characterising the detailed structure of the important worm glycans, we want to understand more about how these interactions take place.
Our analytical method of choice to derive the glycan structures is mass spectrometry (see box), as it is exquisitely sensitive (data can be obtained from very tiny amounts of material, such as 1 femtomole = 1 billionth of a millionth (10-15) of a mole), and it can be used to study very complex mixtures. In a mass spectrometry experiment, energy is transferred to the purified worm glycans, for example by pulsing them with a laser beam. This energy transfer makes them ionised and charged. Once they have a charge, they can be made to ‘fly’ through the analytical section of the mass spectrometer. There, the different glycans are separated by their mass-to-charge ratio. From this information, the structure of the glycans can be deduced in terms of their monosaccharide composition and in terms of how they are linked together. Our mass spectrometry analyses revealed that both the cercarial and egg secretions contain very similar, highly immunogenic glycan structures.
In the case of the non-motile egg, it is as though the egg were trying to attract attention to itself. This led us to think that the parasite egg actually relies on the host immune response, which produces factors such as proteases, to help it escape from the blood vessels to the gut lumen or the bladder. Why the eggs would take the risk of being attacked by the immune system – when they have their own proteases – remains unclear.
In the case of the mobile cercaria, we propose a ‘smokescreen hypothesis’: we think that cercaria ‘deliberately’ attract attention to their secreted glycoproteins to distract the host’s immune response away from protein targets of the larva which are more vital to its survival. Armed with a detailed knowledge of the parasite glycan structures, we hope to design new anti-parasite drugs or vaccines in the future.
Mass spectrometry
Mass spectrometry is an analytical method used to determine the elemental composition of a sample or molecule. It can be used for both qualitative and quantitative measurements. Not only is it an important method for protein analysis, it is also widely used in space missions to characterise the composition of heavenly bodies. The principle consists of ionising the molecules or molecule fragments in the sample and then measuring their mass-to-charge ratios.
The machine used for this method, a mass spectrometer, is generally composed of three sections (see image below):
- The ion source, in which the sample is split into gas phase ions.
- The mass analyser, where electromagnetic fields are applied to separate the ions by their mass-to-charge ratio. These fields exert forces on the ions; the electric field may speed up or slow down a charged particle, and its direction may be altered by the magnetic field. The magnitude of the deflection of the moving ion’s trajectory depends on its mass-to-charge ratio: according to Newton’s second law of motion, lighter ions are deflected by the magnetic force more than heavier ions.
- The detector, which records and quantifies the ions’ mass-to-charge ratio.
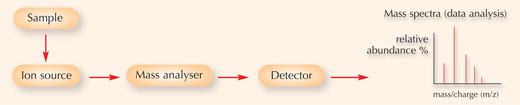
This information is then used to determine the chemical element composition of the original sample.
Acknowledgements
This work was funded by the Biotechnology and Biological Sciences Research Council (BBSRC) and the Wellcome Trust, with additional funds from the UNDP/World Bank/World Health Organization Special Programme for Research and Training in Tropical Diseases.
References
- Kariuki TM, Farah IO, Wilson RA, Coulson PS (2008) Antibodies elicited by the secretions from schistosome cercariae and eggs are predominantly against glycan epitopes. Parasite Immunology 30(10): 554-62. doi: 10.1074/mcp.M700004-MCP200
Resources
- Jang-Lee J, Curwen RS, Ashton PD, Tissot B, Mathieson W, Panico M, Dell A, Wilson RA, Haslam SM (2007) Glycomics analysis of Schistosoma mansoni egg and cercarial secretions. Molecular and Cellular Proteomics 6: 1485-1499. doi:10.1074/mcp.M700004-MCP200
- For more information about schistosomiasis, see: www.york.ac.uk/res/schisto/background.htm
- For more information about mass spectrometry, see: www3.imperial.ac.uk/lifesciences/research/molecularbiosciences/massspec





