What do we know about climate? The evidence for climate change Understand article
In the first of two articles, climate researcher Rasmus Benestad from the Norwegian Meteorological Institute examines the evidence for climate change.
In recent years, climate change has become a considerable international political issue, involving the Kyoto Protocol and discussions at high-level meetings such as the G8 summits. The recent fourth assessment report (AR4) of the Intergovernmental Panel on Climate Change (IPCC) states that the global warming recorded over recent decades is very likely caused by human activity (with more than 90% probability).
International scientific societies have called for actions to mitigate global warming caused by emissions of greenhouse gases such as CO2; the IPCC and former US Vice President Al Gore shared the Nobel Peace Prize 2007 for disseminating knowledge about man-made climate change; there was as an Academy Award for Gore’s documentary An Inconvenient Truth; and the Stern Review Report views climate change as a major challenge for the world.
At the same time, the media has provided reports giving the impression that the science behind anthropogenic (man-made) global warming is controversial, and debates now range from scientific issues concerning climate change to questions about how to deal with climate change. So what do we really know about climate? Here we examine the evidence and arguments concerning climate and climate change.
The history of climate research
It is useful to put the notion of climate change into a historical context, and note that it has been a topic of debate before. In the late 19th century, scholars discussed whether the local climate was affected by deforestation, or whether sunspots had any influence on the weather. At the end of the 19th century, the Mount Haldde aurora observatory in northern Norway was founded – based partly on the belief that Aurora Borealis, or Northern Lights, may affect the weather.
Some scholars believed that climate follows certain cycles, and by looking to the past, one could see patterns that could be used to predict the future. Around the same time, there were also scholars who studied the effects of increases in the atmospheric concentrations of CO2. They thought that variations in the CO2 concentrations might help explain the pronounced temperature variations associated with the ice ages, since the effects of changes in the Earth’s orbit around the Sun were considered too weak to be involved. ‘The Discovery of Global Warming’w1 is an excellent website on these historical aspects.
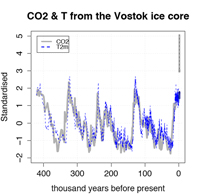
reconstructions from the Vostok
ice core. On the far right are the
present CO2 concentrations.
Click on image to enlarge.
Image courtesy of Rasmus Benestad
How do we know that there were ice ages in the first place? The theory of glacial and interglacial periods was inspired by early geological findings and other geological evidence. Geologists asked what had carved the valleys in the mountain ranges, caused rock scouring and glacial moraines, and came up with the only plausible explanation: ice. There was also palaeontological evidence, such as remainders of vegetation and animals associated with warmer climates, which suggested that the local climate had been warmer, and fossils suggesting that previous sea levels were different from current levels.
More recently, firmer chemical evidence has been drawn from ice cores drilled in the Antarctic and Greenland ice sheets (see graph). Air bubbles trapped in the ice, at depths representing the time when the layer of ice was formed, provide clues about the temperatures and the concentrations of atmospheric trace gases.
Our understanding of the ice ages relies on two pillars: the Milankovitch theory explaining how the timing of the ice ages was related to changes in Earth’s orbit around the Sun, and Arrhenius’ explanation of how variations in CO2 levels could influence the mean global temperature.
The debates continue; but in hindsight, were the scholars of the past wrong about the climate?
Facts about our climate

board the International Space
Station
Image courtesy of NASA
The natural variations tell us that our climate is sensitive to changes in external conditions, such as the energy that the Earth receives from the Sun. We also know that the average surface temperature of Earth should be lower than it is, if we just consider the balance between the energy received from the Sun and the heat that escapes into space.
There is already a natural greenhouse effect keeping the Earth’s surface ~30ºC warmer than the energy balance would suggest. How do we know this? This notion is based mostly on the laws of physics (which have been successful in all other circumstances), which state that our planet receives energy only from the Sun in the form of light, measured by the solar constant of ~1370 W/m2. If the planet doesn’t warm up or cool down over time, then the energy input from the Sun must be in balance, or equilibrium, with the amount of energy that Earth loses to the Universe.
This energy loss follows a well-established physical law (Stefan-Boltzmann law), which has been confirmed by laboratory experiments and states that heat loss (black-body radiation) is temperature-dependent. It is also important to take into consideration the proportion of light reflected from the planet (from clouds and ice, for example).
The energy balance can be used to predict the surface temperature of other planets in our Solar System, and the verification by empirical measurements hence constitutes a scientific test. The intensity of the sunlight is easily calculated from the solar constant and the planet’s distance from the Sun.
In general, the surface temperature of planets decreases as the distance from the Sun increases, as expected from a radiative energy balance. But some planets – such as Earth – have higher surface temperatures than those predicted by the pure radiative energy balance model. These are the planets with an atmosphere that contains greenhouse gases (GHGs). For example, the surface of Venus, which has an atmosphere containing GHGs, is hotter than that of Mercury, which is closer to the Sun but has no GHG-containing atmosphere.

Image courtesy of NASA
Laboratory experiments confirm the greenhouse gas properties of CO2. GHGs such as CO2 have one important physical property: they are transparent in visible light but opaque in infra-red light (heat radiation).
The greenhouse effect can also be understood from the perspective of quantum physics, where photons are absorbed by increasing the rotation or vibration of certain molecules.
When the molecules subsequently lose the energy received by the photons, they radiate about half of the energy back to the ground. When this radiation is added to the sunlight received from the Sun, the net effect is a surface warming.
In summary, the theory behind the greenhouse effect is solid and based on both empirical evidence and theoretical physics. It has also been established from several different lines of evidence that Earth’s climate has undergone variations in the past. These variations can be explained in terms of changes in factors that affect Earth’s energy balance.
Web References
- w1 – The Discovery of Global Warming: www.aip.org/history/climate/index.html
Resources
- Climate Change Education is a website for students, teachers and many more: http://climatechangeeducation.org
- RealClimate is a commentary site on climate science by active climate scientists for the interested public and journalists. It aims to provide a quick response to developing stories and the context sometimes missing in mainstream commentary. The discussion on RealClimate is restricted to scientific topics and does not include any political or economic implications of the science. See www.RealClimate.org
Review
Everyone has heard about climate change. The subject is frequently addressed by the media, but the information given is often incomplete or biased by political views. As science teachers, we have to provide students with correct information and instruments to develop critical attitudes based on facts and aimed at active citizenship.
Rasmus Benestad’s two-part article is very useful because it presents the topic clearly and objectively, addressing its history, available evidence, anthropogenic contribution and controversial issues. Moreover it gives the reader valuable web references for further deepening his or her knowledge of the subject.
I recommend this article to science teachers who are willing to update their knowledge and to secondary-school students interested in the facts and scientific evidence at the heart of this debate. The material is also particularly suitable for classroom discussion and for an interdisciplinary approach to environmental education in secondary schools.
Giulia Realdon, Italy





