Bringing particle physics to life: build your own cloud chamber Teach article
Particle physics is often seen as something only for huge research institutes, out of reach of the general public. Francisco Barradas-Solas and Paloma Alameda-Meléndez demonstrate how – with the aid of a homemade particle detector – you can dispel this myth by bringing particle physics to life…

source emit in a flower-like
pattern at the center of a
continuous cloud chamber.
The particles are made visible
by means of alcohol vapour
diffusing from an area at
room temperature to an area
at -78 °C. This photograph
was taken in 1957. Click to
enlarge image
Public domain image; image
source: Wikimedia Commons
The objective of elementary particle physics is to find the basic building blocks of which everything is made and to investigate the behaviour of these building blocks. Although it can be seen as a cornerstone of science, particle physics is often neglected or poorly understood in schools, partly because it is perceived as unrelated to the things with which we interact on a daily basis. However, particle physicists detect and measure electrons, photons or muons every day with the same confidence with which all of us ‘detect’ cows, tables or aeroplanes. Furthermore, particle detectors (e.g. PET scanners) are routinely used, for example, by medical physicists to detect tumours and monitor the function of internal organs.
Here we demonstrate how to bring particle physics to life in the classroom, using possibly the simplest type of particle detector: a continuously sensitive diffusion cloud chamber. This homemade version consists simply of an airtight fish tank full of air and alcohol vapour, cooled to a very low temperature, which can be used to detect charged particles, particularly cosmic ray muons, if they have enough energy.
Elementary particles
Elementary particles are the simplest elements from which everything is made. They are not just the building blocks of matter and radiation, but also give rise to the interactions between them (for more details of elementary particles, see Landua & Rau, 2008). These particles carry energy and momentum, and can thus be seen by detectors. Strictly speaking, you cannot directly see any particles – instead, their passage through detectors is inferred from the effects they cause, such as ionisation (for charged particles). That is precisely what we do when we observe the condensation trail left in the sky by an aeroplane that we cannot see – and what we can do with our homemade cloud chamber.
The continuously sensitive diffusion cloud chamber

sensitive diffusion cloud
chamber
Image courtesy of Francisco
Barradas Solas
This cloud chamber is basically an airtight container filled with a mixed atmosphere of air and alcohol vapour. Liquid alcohol evaporates from a reservoir and diffuses through the air from the top to the bottom of the chamber. Cooling the base with dry ice (solid carbon dioxide, which is at a constant temperature of around –79 ºC while it sublimates) results in a strong vertical temperature gradient, so that a zone with supersaturated alcohol vapour forms close to the bottom. This sensitive layer is unstable, with more very cold alcohol vapour than it can hold. The process of condensation of vapour into liquid can be triggered by the passage of a charged particle with enough energy to ionise atoms in its path. These ions are the condensation nuclei around which liquid droplets form to make a trail.
Assembly and operation
Materials

chamber. Click to enlarge
image
Image courtesy of Francisco
Barradas Solas
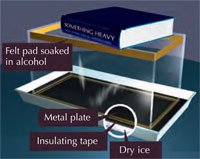
- Straight-sided, clear plastic or glass container (e.g. a fish tank) with a base about 30 cm x 20 cm, and a height around 20 cm (other sizes can be used, but the effects may vary)
- Aluminium sheet (about 1 mm thick, same thickness as the base of the fish tank)
- Shallow tray somewhat larger than the base area of the fish tank
- Two lamps, one of them strong
- Strip of felt (about 3 cm wide and long enough to wrap around the inside of the fish tank, e.g. somewhat more than 1 m long)
- Glue (not alcohol-soluble)
- Black insulating tape or duct tape
- Isopropyl alcohol (isopropanol)
- Dry ice
image
Image prepared by Alberto
Izquierdo; courtesy of Francisco
Barradas Solas
Method
- Glue a strip of felt (the alcohol reservoir) around the insides at the bottom of the fish tank (the body of the cloud chamber). Some felt can be glued to the bottom of the tank, too.
- Cut the aluminium sheet to fit (as closely as possible) the top the fish tank, and cover one side of the sheet with insulating tape, forming a black surface.
- Soak the felt with isopropyl alcohol (but not so much that it drips down the sides of the chamber).
Safety note: Do this in a well-ventilated room and remember that alcohol is flammable. - Turn the fish tank upside-down over the aluminium sheet. Make sure the black side of the sheet faces upwards (to make the particle tracks more visible).
- Use insulating or duct tape to fasten the aluminium sheet to the rim of the fish tank, sealing the chamber so that it is airtight. This is the most critical step and must be carefully done, as the joint will become moist and very cold during operation.
- Make a flat layer of dry ice in the tray and place the chamber on top of it, making sure that its base is horizontal. To ensure good thermal contact between the metal plate and the dry ice, avoid large chunks of dry ice: flat sheets or dust are best, but small grains will do.
Safety note: Dry ice is around –79 ºC and should only be handled using thick gloves. - Keep the top of the chamber warm, for example by shining a lamp onto it. Avoid using the chamber in a cold environment, because this could prevent the correct temperature gradient from forming, meaning no tracks can be seen.
- Leave the chamber undisturbed for about 10 min, until the temperature gradient is established. Shine a bright light through the chamber at a low angle, and look at the bottom of the chamber. At first you should see only an alcohol mist falling, but gradually, charged particle tracks should appear as thread-like condensation in the mist. Note: the tracks are more visible in a darkened room.

in a cloud chamber (thick
and short: alpha particles;
long and thin: beta particles)
Image courtesy of Bionerd;
image source: Wikimedia
Commons
Although any charged particle with enough energy, for example from ambient radioactivity, can leave its trail in the chamber, the majority of the tracks will be made by secondary cosmic rays: particles created when other particles (mostly protons) coming from outer space hit the upper atmosphere. Secondary cosmic rays travel at close to the speed of light and are absorbed by the atmosphere or decay in flight, giving rise to new particles including muons, which can reach the surface of Earth and are easily detected. Muons are charged elementary particles very similar to electrons except for their mass (which is two hundred times larger).

image
Image prepared by Alberto
Izquierdo; courtesy of Francisco
Barradas Solas
What you can do with the chamber?
In order to make the chamber really useful, we cannot limit ourselves to showing it and describing how it works. To support the explanation, we have prepared a short, simply written comic stripw1 (see below), showing how the chamber works and illustrating the origin and composition of cosmic rays through the story of a cosmic proton and its descendants.
We use this chamber at school with our 12- to 16-year-old students as part of an effort to help them see particles as real physical objects. Watching the visible trails left by invisible particles and comparing them to trails left by jet engines (in which much of the same physics is involved) is the first step in a process that we continue by introducing real data and pictures from high-energy physics into otherwise standard exercises and questionsw2, w3 (Cid, 2005; Cid & Ramón, 2009) and that we conclude with another, more complicated, detector for school use: a cosmic-ray scintillation detector which allows students to record and study data by themselves (Barradas-Solas, 2007).

Taken from the cloud chamber
comic strip; image courtesy of
Paloma Alameda-Meléndez
Acknowledgements
The authors would like to thank Dr Eleanor Hayes, Editor-in-Chief of Science in School, for her assistance in giving the final form to this article.
References
- Barradas-Solas F (2007) Giving new life to old equipment. Physics Education 42: 9-11. doi: 10.1088/0031-9120/42/1/F03
To access this article, which is freely available online, visit the website of the Institute of Technical Education, Madrid, Spain (http://palmera.pntic.mec.es) or use the direct link: http://tinyurl.com/y8ssyc5
- Cid R (2005) Contextualized magnetism in secondary school: learning from the LHC (CERN). Physics Education 40: 332-338. doi: 10.1088/0031-9120/40/4/002
- Cid X, Ramón C (2009) Taking energy to the physics classroom from the Large Hadron Collider at CERN. Physics Education 44: 78-83. doi: 10.1088/0031-9120/44/1/011
- Landua R, Rau M (2008) The LHC: a step closer to the Big Bang. Science in School 10: 26-33. scienceinschool.org/2008/issue10/lhcwhy
Web References
- w1 – The comic strip (in English and Spanish) and full assembly instructions (in Spanish) are available from our website: http://palmera.pntic.mec.es/~fbarrada/cc_supp_mat.html
- w2 – See, for instance, the introductory information about the LHC and simple physical calculations which take place in all particle accelerators (Physics at LHC) on the ‘Taking a closer look at LHC’: http://www.lhc-closer.es
- w3 – The CERN website for high-school teachers (http://teachers.web.cern.ch) also includes a gallery of bubble chamber pictures which fits nicely into our project. See the direct link: http://tinyurl.com/yfbv8ls
Resources
- For brief, simple overviews of particle physics aimed at the general public, see:
- Close FE (2004) Particle Physics: A Very Short Introduction. Oxford, UK: Oxford University Press. ISBN: 9780192804341
- The Lawrence Berkeley National Laboratory’s online interactive tour, ‘The Particle Adventure: the Fundamentals of Matter and Force’: www.particleadventure.org
- The virtual visitor centre of the SLAC National Accelerator Laboratory (particularly the sections on theory, detectors and cosmic rays): www2.slac.stanford.edu/vvc
- The CERN website: http://public.web.cern.ch/public/en/Research/Detector-en.html
- For a discussion of how some of the big questions in particle physics will be addressed by CERN’s Large Hadron Collider, see:
- Landua R (2008) The LHC: a look inside. Science in School 10: 34-47. scienceinschool.org/2008/issue10/lhchow
- For more detailed, yet accessible, introductions aimed at people with a scientific education and not afraid of a little mathematics, we recommend:
- Barnett RM et al. (2000) The Charm of Strange Quarks: Mysteries and Revolutions of Particle Physics. New York, NY, USA: AIP Press. ISBN: 0387988971
- Treiman SB (1999) The Odd Quantum. Princeton, NJ, USA: Princeton University Press. ISBN: 0691009260
Treiman’s book is one of the best to begin tackling the subtleties of quantum mechanics in particle physics (which we have avoided in this article), including virtual and unstable particles, and the field / particle relationship.
- To learn more about cosmic rays, see NASA’s Cosmicopia: http://helios.gsfc.nasa.gov/cosmic.html
- We and many others have learned about building cloud chambers from Andy Foland’s cloud chamber page: www.lns.cornell.edu/~adf4/cloud.html
- The American Museum of Natural History’s website includes an illustrated version of the main stages of the assembly of the cloud chamber: www.amnh.org/education/resources/rfl/web/einsteinguide/activities/cloud.html
- It is not easy to explain in detail the processes of supersaturation and track formation or to justify the choice of active liquid (isopropanol, in our case), as they depend in a complicated way on – for example – ionisation energies, vapour pressures, diffusion rates and various engineering aspects of the chamber. If you want to pursue this further, see the supplementary bibliography on our cloud chamber website: http://palmera.pntic.mec.es/~fbarrada/cc_supp_mat.html
- For a Science in School article describing how to measure radioactivity from radon in the home, see:
- Budinich M, Vascotto M (2010) The ‘Radon school survey’: measuring radioactivity at home. Science in School 14: 54-57. scienceinschool.org/2010/issue14/radon
Review
Cosmic rays consist of subatomic particles that come from space and strike Earth’s atmosphere, creating a shower of secondary particles that can be studied at the Earth’s surface. Students in secondary education can usually only read about those particles in books or study them through simulations – although the particles constantly pass through our bodies.
Here, Francisco Barradas-Solas and Paloma Alameda-Meléndez present the idea that cloud chambers can be used by students as an experimental tool, enabling them to conduct their own investigations on radiation. They also provide details about the construction of a cloud chamber, equipment that can be built at school without too much difficulty, which enables students to observe these subatomic particles in the classroom by making their tracks visible.
Vangelis Koltsakis, Greece





