Investigating the causes of schizophrenia Understand article
Laurence Reed and Jackie de Belleroche discuss schizophrenia – and how functional genomics could help to identify its causes.
The experience of schizophrenia

iStockphoto
私
“Don’t you think that Japanese letters – the kanji – are the most extraordinary things in the world? You can just write this symbol, 私 – which means ‘I’ – and it can communicate with people. It tells them where you have been and what you think; some kanji – the powerful ones – can tell them what to think and what to do.
I started to notice this at school – I saw that some kanji were much more beautiful and much more powerful than others. I took down my posters in my room and painted my favourite kanji on the walls – the good ones – not the ones that made me feel afraid. My mother became very upset by this – we argued a lot about this – and tried to paint over them.
I couldn’t go home – my mother had got rid of the good kanji – everywhere I looked there were bad kanji – I even started to be able to hear them. I slept wherever I could – it had to be outside – inside, I felt so trapped. I lost lots of weight and felt very weak. Six months later, I was picked up by the police and they took me to the clinic…”
Introduction to schizophrenia
Schizophrenia affects about 1 in 100 people and is recognised worldwide. In this article, we will discuss what schizophrenia is and what its potential causes are, and then introduce some of our recent research to investigate the combined genetic and environmental influences that can lead to this devastating condition.
The best introduction to schizophrenia is to listen to what the person with a diagnosis of schizophrenia actually says – and that is the introduction we have tried to give here. AB’s account above is fictitious, but it is based upon a real case. The story of each person who suffers from schizophrenia is different, but there are some common and important features, which AB’s case history shows.
The most striking features of schizophrenia are those of psychosis – meaning delusions or hallucinations. Like many words used to describe psychiatric disorders, these terms are often used quite loosely, although they have precise definitions. A delusion is when someone believes very odd ideas for reasons that do not justify the belief. A hallucination is the experience of sensations without any external physical cause for the sensation. For example, in psychosis, these hallucinations are often in the form of ‘voices’, which may tell the person what to do, or may talk about the person, commenting on their actions.
These features tend to appear in adolescence, and tend to be ‘paranoid’ – which simply means that the experiences that a person is having refer in some way to themselves, as if they are in some way special. In AB’s case, he experienced his mother’s attempts to remove the kanji as threatening him directly, so he had to leave home.
Although these symptoms (known as positive symptoms) are striking, they are most often accompanied by negative symptoms – inactivity, inability to relate to people and inability to look after oneself. In AB’s case, he has left school, left home and is walking the streets unable to look after himself. Negative symptoms tend to appear slowly and gradually, and tend to last the lifetime of the sufferer. Whereas current medical treatments work reasonably well with positive symptoms, negative symptoms are more difficult to treat.
Possible causes of schizophrenia
The more we understand about a disorder, the more likely we are to be able to diagnose and treat it effectively. In particular, it is important to understand what causes the disease or contributes to it. How much do we know about the causes of schizophrenia?
Figure 1 (below) shows many of the factors that we know can influence a person’s behaviour, whether or not they are mentally ill. These factors are shown in a hierarchy, or a ladder, starting from the most fundamental individual characteristics of a person through to factors that affect many people.
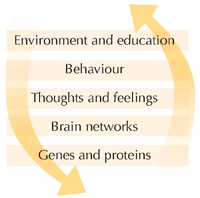
causes of psychiatric
disorders
Image courtesy of Laurence
Reed
At the base of this hierarchy are the genes that a person inherits from their parents. These genes make mRNA that makes proteins that make cells, including networks of nerve cells in our brains. These networks give rise to our thoughts and feelings, and hence to how we behave. The most important point is that this hierarchy doesn’t just work in one direction: the environment can affect how we behave, think and feel, and can produce changes in the brain itself – that is why the arrows go in both directions – and it is not always possible to say which are the most important factors without studying the disorder carefully.
There are a number of clues about possible causes for schizophrenia. It tends to run in families, suggesting a genetic cause. However, it also tends to occur more often in conditions of overcrowding, suggesting that the environment has a role to play. Some drugs such as amphetamines cause symptoms of psychosis. All these different factors may also work together in a complicated way to cause schizophrenia – this is what we have tried to show in Figure 1.
Evidence for a genetic contribution
One of the simplest ways of understanding how our genes contribute to any condition is to examine how that condition is shared between identical twins. If a condition is wholly genetic, then if either identical twin develops the condition, the other twin will develop the condition as well. This is called 100% concordance. If the concordance rate is less than 100% (i.e. the other twin does not always develop the condition), then other factors must contribute to the development of the condition as well. In schizophrenia, twin studies report that concordance rates amongst identical twins worldwide are 41-65%, suggesting that both genetic and non-genetic (environmental) factors play a significant part. Untangling the genetic and environmental factors is not simple, however. Furthermore, extensive DNA analysis suggests that there is no single gene involved in schizophrenia; instead, many genes seem to be linked to the condition, and their effect may be independent or cumulative.
Genetic and environmental interactions
One way of investigating the combined effect of genes and the environment is to study which genes are active (expressed) in the brain regions affected in schizophrenia. Which of our genes are expressed depends, of course, on the genes encoded in our DNA but also on environmental factors. For example, smokers have a higher activity (expression) of genes that encode enzymes involved in metabolising tobacco smoke than non-smokers do. We wanted to see whether gene expression also differed between people with and without schizophrenia.
We can determine the level of gene expression by measuring the amount of mRNA and protein that are produced from particular genes. If a gene is expressed more (or less) in people with schizophrenia than in unaffected people, then the gene may be involved in schizophrenia (either as a cause or a consequence of the disease).
Examining gene expression allows the combined influence of both the genetic effects (encoded in the DNA) and environmental effects (which may modify levels of the expression of some genes) to be investigated. Through recent technological advances, it is now possible to use microarrays, which allow the expression of more than 30 000 gene transcripts from a single individual to be analysed simultaneously (for a classroom activity to simulate microarrays, see Koutsos et al., 2009).

onto a surface. A sample containing genes from an individual with schizophrenia is labelled a certain colour: red,
for example. A second sample from a healthy person is labelled green. The two samples are introduced to the
microarray, where they hybridise with the probe DNA, becoming fixed in place. Genes that are expressed in the
schizophrenic individual appear as red spots, and in the healthy individual as green spots. If they are equally
expressed in both, the result is a yellow spot. Microarrays allow researchers to pin-point the differences in
gene expression levels between diseased and non-diseased individuals.
Click to enlarge image
Image courtesy of Wikimedia Commons / Guillaume Paumier
The genes that are expressed, of course, vary from tissue to tissue – this is one reason why different tissues are different. So how did we know in which regions of the brain to test the gene expression? Previous brain imaging and postmortem examinations have helped to define the main brain regions affected by schizophrenia, for example by comparing brain images of people with and without schizophrenia. One of these key brain regions is the frontal cortex, which is responsible for complex planning; damage to this region results in a loss of motivation, similar to the negative symptoms of schizophrenia.
Based on these previous analyses, therefore, we measured the changes in gene expression in the prefrontal cortex (which is part of the frontal cortex), using post-mortem samples from the brains of 28 patients with schizophrenia (the test group) and 23 healthy people (the control cases) in the UK, matched for age and gender. We also compared our results to those of a similar study using a US database (Glatt et al., 2005).
We found 49 genes that each showed a distinct difference in expression between the test and control groups, a difference of the same direction in the US and UK groups (Maycox et al., 2009). Of these 49 genes, 33 were less active (down-regulated) in patients with schizophrenia than in healthy people, and 16 were more active (up-regulated). Potential confounding factors such as exposure to drugs of abuse and alcohol, smoking or pharmacological treatment were examined, but there was no evidence that the expression of these genes was correlated with such factors.
Which functional pathways are affected?
When we looked at the functions of the 49 genes that were expressed differently in healthy people and those with schizophrenia, we could see that they were involved in biochemical pathways that affected specific functions in the brain: synaptic neurotransmission, signal transduction, cytoskeletal dynamics (see Figure 2, below) and neurodevelopment.
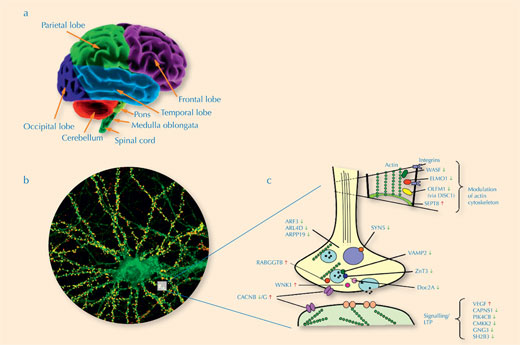
Click to enlarge image
a) We analysed the prefrontal cortex in the frontal lobe (purple)
Image courtesy of www.BrainHealthandPuzzles.com
b) The cellular structure in these brain regions: a neuronal cell body with abundant tree-like branches known as
dendrites, which receive numerous inputs from other neurons. The point of contact between neurons is known
as the synapse (white box)
Image courtesy of www.its.caltech.edu/~mbklab
c) The synapse, including examples of genes that are expressed differently in schizophrenia.
Red arrows indicate up-regulation; green ones indicate down-regulation
Image courtesy of Maycox et al. (2009)
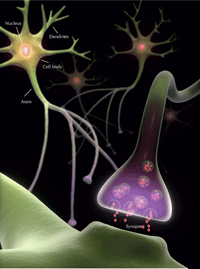
of a neuron, showing the
release of a neurotransmitter
chemical into the synaptic
cleft. The neurotransmitter
interacts with receptors on
the adjacent cell membrane,
propagating the nerve
impulse.
Click to enlarge image
Image courtesy of Animean /
iStockphoto
To understand this, we need to take a closer look at brain cells: neurons. Typically, a neuron consists of a cell body, containing the nucleus; many extensions of the cell body, known as dendrites; and a single extension, called an axon, which may be as long as 1 m (see Figure 3, right).
Neurons are linked together via synapses, allowing electrical nerve impulses to be passed from one neuron to the next: this is known as synaptic transmission and can be initiated by the release of a specialised chemical, a neurotransmitter, into the synapse. The neurotransmitters released by one neuron interact with receptors on either the dendritic spines (swellings on the dendrites) or the cell body of the next neuron. In that second neuron, the chemical signal is converted back into an electrical signal in a process known as signal transduction. The structure of our neurons is, however, not static, but changes according to the signals we receive – our experiences. The structure of the synapse in particular can change in response to the signals it receives – this is how memories are formed, skills are learned and experiences are integrated. These changes reflect the cytoskeletal dynamics of the cell and contribute to the overall process known as synaptic plasticity.
The 49 genes that were expressed differently in people with schizophrenia, therefore, are involved in fundamental brain processes that determine how the cells in the brain respond to external signals and to nerve impulses elsewhere in the brain; these responses include a change in the density of dendritic spines at the synapse. This is consistent with the decreases in synapse density observed microscopically in post-mortem examinations of the brains of people with schizophrenia. Such changes at the synapse are known to be key to the adaptive changes that occur during learning and development – changes known as synaptic plasticity.
The unusual patterns of expression of the genes that we detected in patients with schizophrenia suggest that schizophrenia disrupts the synaptic plasticity in the prefrontal cortex, which we would expect to result in impaired learning and social interaction – features that are indeed associated with schizophrenia. Our results, therefore, suggest that schizophrenia may result from a cumulative effect of multiple factors (genetic and environmental) targeting these functional pathways.
Implications for patients
From these and similar studies, we hope to understand what changes occur at the molecular level in people with schizophrenia. Some of these molecular changes may be common to all patients with schizophrenia, whereas others may correspond to specific features such as cognitive changes that often lead to people with schizophrenia having difficulty concentrating and learning. Fundamentally, this knowledge will provide clues to the processes that take place as schizophrenia develops. If we can understand these processes, this knowledge may help us to diagnose the disease and to design better treatments for patients or for particular groups of patients. Overall, studies such as these provide ‘puzzle pieces’ which will help us to put together the picture of how our genes and environment contribute to the development of schizophrenia.
References
- Glatt et al. (2005) Comparative gene expression analysis of blood and brain provides concurrent validation of SELENBP1 up-regulation in schizophrenia. Proceedings of the National Academy of Sciences USA 102(43): 15533–15538. doi: 10.1073/pnas.0507666102
- Koutsos A, Manaia A, Willingale-Theune J (2009) Fishing for genes: DNA microarrays in the classroom. Science in School 12: 44-49.
- Maycox et al. (2009) Analysis of gene expression in two large schizophrenia cohorts identifies multiple changes associated with nerve terminal function. Molecular Psychiatry 14: 1083-1094. doi: 10.1038/mp.2009.18
Resources
- For more information about schizophrenia, its causes and treatment, see the website of the US National Institute of Mental Health (www.nimh.nih.gov) and the Psychological Information Online website: www.psychologyinfo.com/schizophrenia
- When teaching about schizophrenia, it is important to bear in mind that it may affect one or more of your students. They may be sufferers themselves or they may have a family member who is. Schizophrenia is devastating for the whole family, so do emphasise that help is available. Support groups are available in many countries. For example, the UK organisation Rethink offers support for people with serious mental illnesses and also for their families. To find out more, see: www.rethink.org
- The international, independent non-profit organisation Schizophrenia.com provides similar information and support. See: www.schizophrenia.com
- Written by Christian Seeger, then a university student, the short story ‘I can see things that you can’t see’ describes the effects of childhood schizophrenia. It can be downloaded here (or in the original German, here).
- Edwin H Rydberg’s story ‘Through the illusions’, about the link between schizophrenia and smoking, won the EMBO Science Writing Prize in 2005 and can be downloaded here: www.embo.org/documents/press05/embo_2005_rydberg.pdf
Review
In many peoples’ minds, schizophrenia is equivalent to madness. This image is at least partially caused by ignorance of the nature of the disease. This article provides basic information about schizophrenia and elaborates on the possible causes of this devastating disease.
After an unusual and clever prologue to introduce schizophrenia, the article concentrates on molecular genetic methods that have been used to investigate the causes of schizophrenia. Although the scientific detail and the terminology used might overwhelm a non-scientific audience, advanced high-school biology teachers will most likely find the information highly interesting and useful and could use it in their teaching.
For example, the article could be used in a lesson on diseases that are caused, at least in part, by genetic factors. Some knowledge of molecular genetics would be required. If the students want to investigate the subject further, the author provides references and suggests resources.
Example comprehension questions include:
- What criteria can be used to recognise a person with schizophrenia?
- What are the most common symptoms of schizophrenia? Which are classified as positive and which as negative?
- Describe a method that can be used to find out if a disease is genetic or not, and if it is, to what degree.
- Explain what you have learned about the molecular genetic basis of the causes of schizophrenia.
Michalis Hadjimarcou, Cyprus





