Supporting materials
Download
Download this article as a PDF

The following was adapted from an article in the BMJ and explains why care must be taken in interpreting test results.
A negative result from a medical test means you definitely don’t have the condition, right? Wrong: it depends on the false negative rate of the test and on your individual risk.
Across the world, there has been a clamour for COVID-19 testing, with the director general of the World Health Organization encouraging countries to “test, test, test”. The availability of the complete genome of COVID-19 early in the epidemic facilitated the development of tests to detect viral RNA.[1] Multiple assays with different gene targets have been developed using reverse-transcriptase polymerase chain reaction (RT-PCR).[2] These viral RNA tests, also known as PCR tests, use samples obtained via swabs to detect current infections. See the additional material to learn more about how COVID-19 tests work.
Testing for COVID-19 aims to identify infected individuals so they can isolate and reduce spreading.[3] It also allows contact tracing for exposed individuals,[4] and provides information on regional and national rates of infection to inform public health interventions. However, there is often uncertainty on how to use the test results to make good decisions.
No clinical test gives results that are 100% accurate. Tests can give a false positives (where the test result is positive but the person tested does not have the disease) or a false negatives (where someone who has the disease gets a negative test result). It is thus important to know the ‘sensitivity’ and ‘specificity’ of a particular test.
Sensitivity refers to the true positive rate (i.e., the proportion of people with the disease whose test result is positive). Specificity refers to the true negative rate (i.e., the proportion of people without the disease who test negative). These terms, which can be confusing, are summarized in Table 1.
| Accuracy measure | Definition | Formula |
|---|---|---|
| True positives (TP) | People with the disease who have a positive test result | TP |
| True negatives (TN) | People without the disease who have a negative test result | TN |
| False positives (FP) | People without the disease who have a positive test result | FP |
| False negatives (FN) | People with the disease who have a negative test result | FN |
| True positive rate (TPR) | Sensitivity: proportion of people with the disease who have a positive test result | TP/(TP+FN) |
| True negative rate (TNR) | Specificity: proportion of people without the disease who have a negative test result | TN/(FP+TN) |
| False negative rate (FNR) | Proportion of people with the disease who have negative test result | FN/(TP+FN) |
| False positive rate (FPR) | Proportion of people without the disease who have positive test result | FP/(FP+TN) |
From the formulae in the table, we can see that sensitivity and specificity are closely related to the false negative and false positive rates. If a test has a sensitivity of 70%, this means that the false negative rate is 100 – 70 = 30%. If its specificity is 95%, the false positive rate is 100 – 95 = 5%.
So, how accurate are viral RNA/PCR swab tests for COVID-19? Studies have shown that the accuracy varies depending on several factors: the stage of disease (which affects the number of virus particles in the body and thus how easy they are to detect),[6] the site on the body from which the sample is taken, and on the quality of the sampling. In one study, the sensitivity of viral RNA/PCR swab tests in 205 patients was 63% for nasal swabs, but only 32% for throat swabs.[7] Although figures do vary between studies, in this article we use the approximate numbers of 70% for sensitivity and 95% for specificity, which are in line with current estimates.[8]
Interpretation of a test result depends not only on the accuracy of the test itself, but also on the pre-test probability of disease. The pre-test probability is simply the likelihood of someone having the disease before they have been tested, which depends on factors including their symptoms and on the overall prevalence of the disease.
When people fail to use the pre-test probability in interpreting a test result and only focus on the test result itself, they are missing a vital piece of information. In fact, this is a logical fallacy called ‘base-rate neglect’, which is very common in interpreting statistics. Here, the pre-test probability is the base rate.
Doctors estimate pre-test probabilities unconsciously in everyday practice, and they use these estimates in diagnosing patients. However, faced with a new and unfamiliar disease such as COVID-19, such intuitive estimation is less reliable, so the clinician will use specific information to estimate the pre-test probability, including:
The effect of pre-test probabilities on what a test result means in practice is demonstrated in the case below (see text box). As this example makes clear, a single negative test result is not reliable if the pre-test probability is high.
In the COVID-19 peak of January 2021, a 52 year old family doctor in London develops a cough, intermittent fever, and feels unwell. On day two of her illness, she has a swab test for COVID-19, which is negative. Her cough and fever persist, but she feels well enough to return to work. What should she do?
The pre-test probability is high in someone like this doctor, who has symptoms typical of COVID-19, a risk of exposure through her occupation, and who works in a high-prevalence region (London). We might estimate her pre-test probability of having COVID-19 at 80%. If she has a viral RNA/PCR test and receives a negative result, she still has a 56% chance of having COVID-19. This means that even with a negative test result, this doctor is far more likely than not to have the disease. If this doctor were to return to work, there is a high risk she could infect her patients and colleagues. It is therefore important for her to self-isolate, even though her test results are negative. In this situation, negative test results can be very misleading.
This case illustrates the risk of base-rate neglect: it can be tempting to put too much trust in the results of an ‘objective’ test, and to ignore what the symptoms and local infection rates indicate.
This idea that the pre-test probability can affect the reliability of the result can seem a bit counterintuitive, but the following infographic (figure 1) illustrates this effect. It shows the outcomes when 100 people with a pre-test probability of having COVID-19 of 80% are tested (using a test with 70% sensitivity and 95% specificity). This pre-test probability is realistic for people who have symptoms and who may already have been exposed to infection.
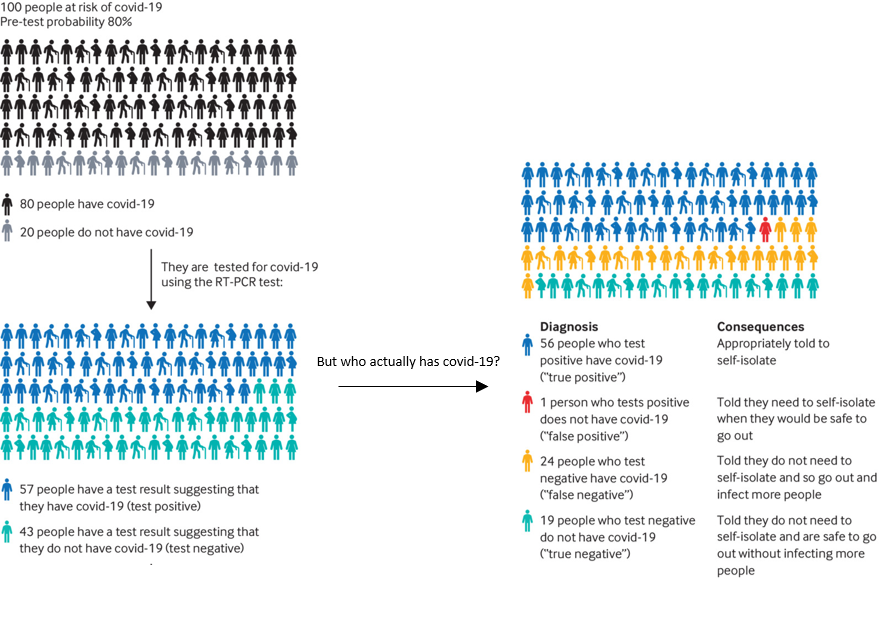
In this example, the number of false positives is very low: just one person in 100. But the number of false negatives is much higher, at 24 in 100. The danger is that these people may assume, based solely on their test result, that they do not need to self-isolate, and so they go out and infect more people.
This interactive infographic shows how the numbers of true and false positives and negatives are affected by the pre-test probability. You can change the pre-test probability and see how the test outcomes change. You can also change the sensitivity and specificity values to see how these affect the outcomes.
While positive tests for COVID-19 are clinically useful, negative tests need to be interpreted with caution, taking into account the pre-test probability of disease. Because of the relatively high false negative rate of the PCR/viral RNA tests (and the more recent ‘lateral flow’ tests, which have an even higher false negative rate), they carry substantial risks: patients may be moved into non-COVID-19 wards, leading to the spread of hospital-acquired infection;[12] carers could spread the infection to vulnerable dependents; and healthcare workers risk spreading COVID-19 to multiple vulnerable individuals. Everyone with a single negative test but with symptoms of COVID-19 should be advised to self-isolate, in keeping with the guidelines for suspected COVID-19.
This article was adapted (with permission) by Susan Watt from an article originally published in the BMJ: Watson J, Whiting FP, Brush JE (2020) Interpreting a covid-19 test result. BMJ 369:m1808. doi: 10.1136/bmj.m1808
[1] Corman VM et al. (2020) Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eur Surveill 25:23–30. doi:10.2807/1560-7917.ES.2020.25.3.2000045
[2] Vogels CBF et al. (2020) Analytical sensitivity and efficiency comparisons of SARS-COV-2 qRT-PCR assays. medRxiv 20048108. doi:10.1101/2020.03.30.20048108
[3] Day M (2020) Covid-19: identifying and isolating asymptomatic people helped eliminate virus in Italian village. BMJ 368:m1165. doi:10.1136/bmj.m1165
[4] Pollock AM et al. (2020) Covid-19: why is the UK government ignoring WHO’s advice? BMJ 368:m1284. doi:10.1136/bmj.m1284
[5] Watson J, Whiting FP, Brush JE (2020) Interpreting a covid-19 test result. BMJ 369:m1808 data supplement: https://www.bmj.com/content/bmj/suppl/2020/05/12/bmj.m1808.DC1/watj056527.ww1.pdf
[6] Sethuraman N et al. (2020) Interpreting diagnostic tests for SARS-CoV-2. JAMA 323:2249-2251. doi:10.1001/jama.2020.8259
[7] Wang W et al. (2020) Detection of SARS-CoV-2 in different types of clinical specimens JAMA 323:1843–1844. doi:10.1001/jama.2020.3786
[8] Woloshin S, Patel N, Kesselheim AS (2020) False Negative Tests for SARS-CoV-2 Infection — Challenges and Implications. N Engl J Med 383:e38. doi:10.1056/NEJMp2015897
[9] Worldometer Covid-19 Coronavirus pandemic data: https://www.worldometers.info/coronavirus/#countries
[10] Public Health England COVID-19: track coronavirus cases: https://www.gov.uk/government/publications/covid-19-track-coronavirus-cases
[11] Centre for Evidence-Based Medicine COVID-19 signs and symptoms tracker: https://www.cebm.net/covid-19/covid-19-signs-and-symptoms-tracker/
[12] Nacoti M et al. (2020) At the epicenter of the covid-19 pandemic and humanitarian crises in Italy: changing perspectives on preparation and mitigation. NEJM Catalyst doi:10.1056/CAT.20.0080
The current pandemic has introduced us all to the new concept of rapid disease testing and quarantine periods. Rolling out accessible tests has meant that we can now be sure if we have got COVID-19, a positive result, but not so sure if we have a result that is negative for the virus. Interpreting negative results is a tricky business and requires local disease information as well as a mathematically researched model. This article focuses on interpreting negative tests and can be used to show people how difficult that can be. It can be used to stimulate discussions on how valuable tests are even if you feel well, how signs and symptoms of disease can be very similar and difficult to diagnose accurately in the absence of testing. They could role play doctor and patient to explain why someone with a negative Covid test who has been in direct contact with a known Covid positive patient should self-isolate. They could explain how a pre-test probability can alter the likelihood of false negative results. Students could have a discussion on the ethics and reliability of testing for all regardless of symptoms, looking at areas where that has occurred and the impact it has had. For advanced biology students, they could research viral replication and the technique of the polymerase chain reaction in the detection of RNA viruses and relate this to viral load.
Dr Shelley Goodman, Lecturer in Applied Science, UK
Download this article as a PDF