Cancer stem cells – hope for the future? Understand article
Cancer and stem cells are both topical issues. But have you heard of cancer stem cells? As Massimiliano Mazza explains, this concept may revolutionise the treatment of cancer.
Cancer: an evolving concept

the world’s oldest surviving
surgical document, was
written in hieratic script in
ancient Egypt around 1600
BC. It is believed to be a copy
of an older work attributed
to Imhotep. Plate 6 and 7 of
the papyrus, pictured here,
discuss facial trauma. Click
on image to enlarge
Public domain image; image
source: Wikimedia Commons
If thou examinest a man having tumours on his breast, (and) thou findest that swelling have spread over his breast (…) thou shouldst say concerning him: “One having tumours. An ailment against which I will fight.”
This first known reference to a tumour, on an Egyptian papyrusw1, is attributed to Imhotep – vizier, architect, physician and astronomer to Pharaoh Djoser, in 2500 BC.

(1821-1902), referred to
as ‘the father of modern
pathology’
Image courtesy of the National
Institutes of Health; image
source: Wikimedia Commons
A tumour (from Latin tumor, meaning swelling) is a tissue mass caused by the abnormal proliferation of cells, but it is not always associated with cancer. Tumours that are confined to a specific region of the body are known as benign – they may cause health problems but are not necessarily life-threatening.
In contrast, a tumour that spreads within the body, invading adjacent tissues and often metastasising (spreading to tissues and organs elsewhere), is called malignant; this is cancer. Although humans have been aware of cancer for more than 4000 years, our understanding of it has only improved significantly in the past 200 years.
An important contribution to the study of cancer was made in the 19th century by German physiologist Johannes Müller and his student Rudolf Virchow. Benefitting from recent advances in microscopy, they understood that cancer is an aberration of cell behaviour, rather than being generated de novo. Virchow was the first to realise that this happens through a combination of genetic predisposition and chronic irritation, such as that caused by smoking or ultraviolet radiation. As we know today, cancer begins with a single cell that loses control over its growth as it accumulates genetic mutations – both spontaneously and as a result of environmental factors – sometimes over a period of many years.
Cancer heterogeneity and cancer stem cells
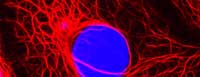
Image courtesy of Lutz
Langbein, German Cancer
Research Center (DKFZ)
The microscopic dissection of tumours in the 19th century also revealed that the morphology of even a single tumour may be remarkably heterogeneous. A tumour always originates from a single cell, so how can its daughter cells vary so much? There are two reasons: first, as they divide, the cancer cells accumulate further mutations that make them genetically different from one another; and second, the microenvironment varies across the tumour and this influences how each cancer cell behaves.
Let us take a look at the genetics first. In certain types of skin cancer, for example, an epithelial cell accumulates mutations, often over many years, and becomes cancerous – proliferating to form a group of epithelial cancer cells. Just like a population of animals, this population of cells evolves as it accumulates further mutations. Those mutations that give a cell a competitive advantage over other cells will result in a clone of similar cells, whereas other mutations will be disadvantageous and may even kill off the cell. By the time a tumour is detected, the different clones of cells that make up the lump may have become distant relatives.
The second influence is the microenvironment of a cancer cell: the cells, structures and molecules around it. As the epithelial cancer cells proliferate and the tumour grows, it incorporates cells from the surrounding tissue, until it consists not only of cancer cells but also of many different types of non-cancerous cells. These can include supporting cells of the connective tissue, blood vessels that supply the tumour with nutrients, and immune cells that enter the tumour from the bloodstream and provide it with cell-signalling molecules and soluble factors. Plus there are normal epithelial cells that are not cancerous, along with the extracellular matrix – structural protein fibres that hold the cells together – and the fibroblasts that synthesise it. A tumour is therefore a complex tissue.
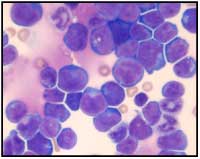
T-cell acute
lymphoblastic eukaemia
Image courtesy of Hind
Medyouf, German Cancer
Research Center (DKFZ)
Is this the whole story of why cancer cells vary so much within a single tumour? Is this variation solely due to the mutations that the rapidly dividing cancer cells inherit and accumulate, plus the influence of the tumour’s microenvironment? According to the stochastic model of cancer formation, it is. This model predicts that all cancer cells within a tumour are equal in their ability to initiate a new tumour if transplanted into a suitable host (Figure 1).
Experiments in the mid-20th century, however, showed that although a single cell can initiate a tumour, it has to be the right cell; this suggested that there are different types of cancer cells within a tumour, each with diverse roles. Indeed, in the 1960s, two distinct groups of cancer cells were identified among tumour cells of leukaemia patients: a larger subset that divided every 24 hours, and a smaller, slow-cycling subset that did not proliferate for weeks or months.
To account for these findings, a second model – the hierarchical or cancer stem cell model – was proposed, according to which the cancer cells are hierarchically organised, as are the cells of normal tissues. The model predicts at least two distinct subpopulations: a small population of cancer stem cells (CSCs) that divide slowly and are responsible for maintaining the tumour, and the vast majority of other cancer cells, which divide more quickly and are not capable of initiating a new tumour (Figure 1). The name ‘cancer stem cells’ was chosen because, like stem cells in normal body tissues, they can either produce more cells like themselves, or differentiate to form different cell types.
Scientists are still debating which of the two models (stochastic or hierarchical) best fits the experimental results, but there is mounting evidence from studies of several cancer types that a small subset of cells are responsible for tumour maintenance and initiation. If so, these are CSCs.
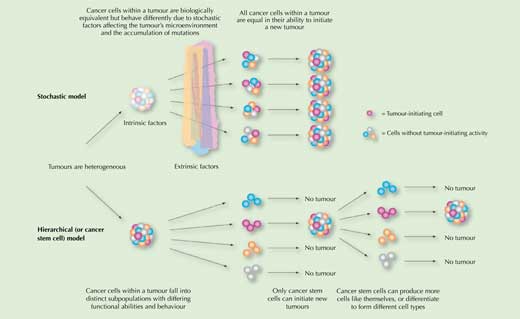
Image reproduced and edited courtesy of American Society of Hematology, from Dick JE (2008) Stem cell concepts renew cancer research. Blood 112(13): 4793-4807. doi: 10.1182/blood-2008-08-077941; permission conveyed through Copyright Clearance Center, Inc
Cancer stem cells in cancer therapy

Rembrandt Harmenszoon van
Rijn (1606–1669). The model
is believed to be Rembrandt’s
second wife Hendrickje, and
the dimpled discolouration
on her left breast is thought
to be breast cancer
Image courtesy of The Yorck
Project: 10.000 Meisterwerke
der Malerei; image source:
Wikipedia
Currently, despite treatment with chemo- and radiotherapy, many cancers recur and metastasise to other organs – these are the circumstances of the majority of cancer patients that die. Why is this? The reason could be CSCs. Because of their slower cell cycle, they might survive the treatment, allowing the tumour to regrow later.
mammograph
Image courtesy of the National
Institutes of Health
Conventional chemo- and radiotherapies for cancer specifically target fast-dividing cells, because most cancer cells proliferate much faster than normal cells. The treatments are designed not to harm slowly dividing cells, such as normal stem cells and most other cells in our body. After patients with acute myeloid leukaemia are treated with these therapies, their haematopoietic (blood-forming) stem cells begin to proliferate to replenish the pool of blood cells. However, scientists have found that the CSCs that have survived start proliferating then too, regenerating the bulk of the tumour with cells even more aggressive and treatment-resistant than before.
The obvious solution, then, would be to develop therapies to specifically target CSCs. This is not easy though, because scientists are still struggling to find reliable methods to detect which cells in a tumour are the CSCs. The idea, however, is for those types of cancer where the CSC model holds true – such as brain, breast, colon, ovarian, pancreatic and prostate cancers – to combine therapeutic strategies that target the fast-dividing cells with strategies that make the slowly dividing CSCs proliferate faster, so that they can be killed off, too.
Over the next few years, scientists hope to find out whether the CSC theory is a real breakthrough for cancer treatment, or if a more accurate model of cancer is still required.
Web References
- w1 – For translations of the Edwin Smith papyrus in English and German, see: www.touregypt.net/edwinsmithsurgical.htm and www.medizinische-papyri.de/PapyrusSmith
Resources
- The University of Rochester Medical Center has produced a lesson plan for an upper secondary-school activity on cancer stem cells. See the university website (www.rochester.edu) or use the direct link: http://tinyurl.com/633jfds
- Harvard scientists have recently discovered that CSCs may be generated from other cancer (non-stem) cells – so that eradicating the CSCs would not eradicate the cancer. See ‘Cancer stem cells made, not born’ on the university website (www.harvard.edu) or use the direct link: http://tinyurl.com/6h9fyng
- Nature has compiled a resource section on CSCs, including a podcast, links and current research articles. See: www.nature.com/nature/focus/cancerstemcells
- For a teaching activity to spot cancer mutations, see:
- Communication and Public Engagement team (2010) Can you spot a cancer mutation? Science in School 16: 39-44. www.scienceinschool.org/2010/issue16/cancer
- Joan Massagué is an inspiring cancer researcher. For an interview with him, see:
- Sherwood S (2008) On the trail of a cure for cancer. Science in School 8: 56-59. www.scienceinschool.org/2008/issue8/joanmassague
Review
This article puts forward the current theory that there are cancer stem cells in much the same way as there are other stem cells. The article could make a good basis for a general discussion of the topic of cancer, including cancer research and treatments, diagnosis and survival rates. Why are some cancers more treatable than others? Differences between bone marrow transplants, chemotherapy, immunotherapy, radiotherapy and vaccination can be researched and discussed. Why is there no vaccine for cancer? How does the vaccine for human papilloma virus work to prevent some cervical cancers (it has to be remembered that this is not a vaccine against the cancer itself)? Some cancers involve chromosomal rearrangements – this would lead on to the topic of cytogenetics. Most students have encountered someone with cancer and they will be able to engage with the topic.
Shelley Goodman, UK





