Winning an Oscar in immunology Understand article
Have you ever wondered what it is that scientists get so excited about? Ana de Barros from the Instituto de Medicina Molecular in Lisbon, Portugal, shares with us the excitement of researching the immune system.

Image courtesy of Ana de Barros
When my phone rang and I was told that our article was to be published in Nature Immunology (Ribot et al, 2009), I just couldn’t stay put. But even though a few people around me understood my hysterics, I had to explain to many others what this really meant for us: “It’s like an actor receiving an Oscar!”
Our particular Oscar was not for best film, actor or soundtrack, but for a discovery in immunology. Having our work published in this prestigious journal meant that fellow scientists had deemed it noteworthy, and that others would now be able to learn from it and build upon it towards the common goals of understanding how our bodies fight disease and subsequently improving human medicine. Sitting on my shelf, ‘our’ issue may look like just another magazine, but it means as much to me as a trophy in a display cabinet.
The subject of our research is the development of a type of white blood cell, called a T lymphocyte or T cell, in the thymus. T lymphocytes are distinguished from other lymphocytes by a molecule on their surface called a T-cell receptor, which recognises the antigen carried by a specific invader, such as a bacterium or virus.
of a lithographic plate from the
20th US edition of Gray’s Anatomy
of the Human Body, originally
published in 1918
Public domain image; image souce
: Wikimedia Commons
T cells are generally considered part of the adaptive immune response, because they not only eliminate an invader (and the cells it has invaded), but also produce memory cells. As their name implies, these memory cells ‘remember’ that invader for as long as the animal lives, so this immune response adapts very quickly: if that same invader strikes again, the memory cells will recognise and destroy it. This adaptive immune response, which only vertebrates possess, is activated by the innate immune response, the non-specific defence system found in all classes of animal and plant life.
The innate immune response is the first line of defence: the initial reaction to (and attempted elimination of) invaders. Unlike the adaptive immune response, the innate immune response does not confer long-lasting or protective immunity, but it is a swifter and more general response.
Our work focuses on a specific type of T cell, the gamma delta (??) T cell, which is seen as an evolutionary bridge between the adaptive and the innate immune responses. ?? T cells form subtypes with different functions, depending on what tissue they are found in and which specific T-cell receptor they bear. Even though there is a growing consensus about the importance of ?? T cells, little is known about their behaviour or development, and scientists haven’t yet found any molecular markers that they can use to identify the different subtypes.
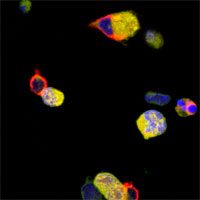
T cells (in red) attacking tumour
cells (in yellow)
Image courtesy of
Bruno Silva-Santos’ lab
In our research, we looked at a protein that we suspected might be a potential marker for one subtype of ?? T cells. This protein, called CD27, is a membrane receptor involved in cellular communication.
We found that our suspicions were well founded: CD27 can be used to identify a particular subtype of γδ T cell. But what was most interesting was that we discovered that the presence of this protein doesn’t merely characterise this subtype of γδ T cell, it actually drives its formation. Within the thymus, the precursors of γδ T cells that mature in the presence of CD27 (CD27+) produce a cytokine called interferon-gamma (IFNγ), whereas cells that mature without CD27 (CD27-) produce primarily a different cytokine, interleukin-17 (IL-17). The presence or absence of the cytokine CD27 thus defines which subtype a gd T cell will belong to (see top diagram below).
The two cytokines IFNγ and IL-17 play an important role in the immune response, with very distinct consequences: those T cells producing IFNγ will play a crucial role in fighting viruses and tumours, whereas those producing IL-17 are associated with autoimmune diseases such as multiple sclerosis.
γδ T cells play an important role in initiating a quick immune response to various parasites, including the malaria parasite Plasmodium. In humans, this parasite multiplies in the liver, and then spreads to the blood stream, infecting red blood cells, which it destroys. In severe cases this can clog the blood vessels that carry blood to the brain, causing what is known as cerebral malaria. We were interested in discovering whether the different subtypes of γδ T cells influenced this outcome differently: was one subtype of γδ T cell better at preventing cerebral malaria than the other?
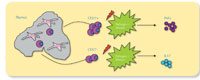
formation of γδ T cells.
Click to enlarge image
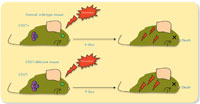
quickly when infected with
Plasmodium than the
CD27-deficient (CD27-) mice did.
Click to enlarge image
Images courtesy of Ana de Barros
To analyse the behaviour of both gd subtypes in the course of infection, we used mice infected with a strain of Plasmodium as a model for what happens in humans. We compared two groups of mice: one group with CD27 in most of their cells (so the majority of their cells produced IFNg), and another genetically engineered group in which none of the maturing γδ T cells had CD27 – which meant that they produced less IFNγ. Both groups of mice were infected with the malaria parasite, and we found that CD27 does indeed seem to influence the development of cerebral malaria.
Mice without CD27 suffered less from cerebral malaria, probably because they produce less IFNγ, which is involved in inflammation (see lower diagram above). Whether this finding could be used in the development of future malaria drugs – perhaps by manipulating the expression of CD27, so that the cells produce lower levels of pro-inflammatory cytokines – remains to be seen.
In short, we were the first to describe a function for CD27 in T-cell precursors in the thymus – and the fact that Nature Immunology decided to publish our article is recognition of just how important these findings are. As our group leader, Bruno Silva-Santos, explains, “We are now investigating the analogous processes in human cells. Our long-term goal is to… be able to manipulate [γδ T cells] for therapeutic purposes such as fighting autoimmune diseases and cancer.”
We still have a lot of work ahead of us, but this recognition of what we have already achieved reminds us that it is all worth it.
Long after I had hung up the phone, then finished explaining my excitement, I still couldn’t stop smiling.
References
- Ribot JC, deBarros A, Pang DJ, Neves JF, Peperzak V, Roberts SJ, Girardi M, Borst J, Hayday AC, Pennington DJ & Silva-Santos B (2009) CD27 is a thymic determinant of the balance between interferon-g and interleukin 17-producing gd T cell subsets. Nature Immunology 10: 427-436. doi:10.1038/ni.1717
Web References
- w1 – Find out more about the Instituto de Medicina Molecular in Lisbon here: www.imm.fm.ul.pt
Resources
- Hodge R (2006) Fighting malaria on a new front. Science in School 1: 72-75.
Review
Immunology is still a fast-moving subject as more is discovered about the involvement of the different types of cells and signalling molecules. Cerebral malaria in humans is currently very difficult to treat. A specific subtype of T cells and the signalling molecule CD27 are involved in the progress of cerebral malaria in mice and this research may have implications for the treatment of this disease in humans. Using this article for background information may stimulate discussion on malarial treatment and eradication, basic immunology, genetic manipulation of mice and whether animal experimentation is right. It could be used for comprehension with further questions set to probe students’ understanding of immunology at the upper secondary level.
Shelley Goodman, UK





