Better milk for cats: immobilised lactase used to make lactose-reduced milk Teach article
Dean Madden from the National Centre for Biotechnology Education (NCBE), University of Reading, UK, suggests an experiment to make lactose-free milk – useful both for cats and for the 75% of the world’s human population that are intolerant to this type of sugar.
Aims
This simple practical investigation introduces students to the principles of digestion and enzyme immobilisation. It can be used as the starting point for other, more advanced activities such as the regulation of lactase production in Escherichia coli (the lac operon), the evolution and social significance of lactose tolerance in humans, and the use of enzymes in food production.
Introduction
Lactase (beta-galactosidase) catalyses the hydrolysis of lactose to glucose and galactose:
Lactose -> D-glucose + beta-D-galactose
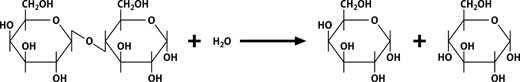
Both of these sugars taste sweeter and are more readily digestible than lactose. Despite their traditional fondness for milk, cats are unable to digest large amounts of lactose. Milk can be treated with the enzyme to make a lactose-reduced milk suitable for cats or for humans who are lactose intolerantw1.
Although the production of a special ‘cat milk’ may seem trivial, an estimated 75?% of the world’s human population are lactose intolerant in adulthood – it is lactose tolerance that is unusual.
Commercially, milk is treated by injecting an enzyme into the carton as UHT milk is packaged, or by using an immobilised enzyme – an enzyme that has been trapped on an inert material so that it can be used repeatedly.
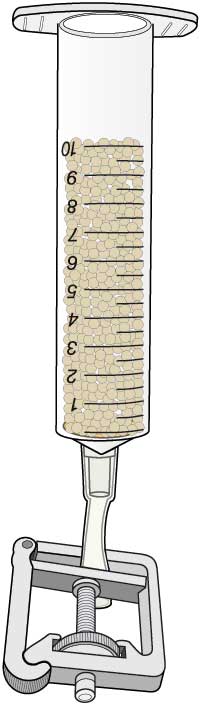
In this activity, students immobilise the lactase in calcium alginate beads held within a small column, over which the milk is passed.
Equipment and materials
Needed by each person or group:
Equipment
- Small piece (about 1 cm2) of nylon gauze, e.g. net curtain
- 10 ml plastic syringe (without a needle)
- 4 mm diameter aquarium airline or silicone tubing, about 7 cm long, to fit syringe
- Aquarium airline tap or adjustable laboratory tubing clip (Hoffman clip)
- Retort stand, boss and clamp (to support enzyme column)
- 2 small beakers (100 ml) or disposable plastic cups
- Tea strainer
- Glass stirring rod
Materials
- 2 ml lactase enzyme (Novozymesw3 Lactozym®)
- 8 ml 2?% sodium alginate solution
- 100 ml 1.5?% calcium chloride solution
- 50 ml milk (not UHT milk)
- Semi-quantitative glucose test strips (e.g. Roche Diabur-Test 5000 or Ames Diastix)
Note: All solutions must be made using distilled or deionised water. Sodium alginate is not readily soluble, and requires both warm water and stirring to dissolve.
Preparation and timing
This activity takes about 40 minutes. The sodium alginate takes some time to dissolve, so the solution is best prepared before the lesson. The immobilised enzyme may be prepared in advance if desired: the beads should be refrigerated, although they will not keep for more than a few days.
Procedure
- Mix the enzyme with the sodium alginate solution, then draw it up into a 10 ml syringe.
- Add the alginate-enzyme mixture a drop at a time from the syringe to the calcium chloride solution and observe the formation of small beads. Do not allow the tip of the syringe to come into contact with the calcium chloride solution, as this will cause the alginate to harden, blocking the outlet. The beads, which contain the enzyme immobilised in a matrix of calcium alginate, should be allowed to harden for a few minutes.
- Attach a short length of tubing to the tip of a syringe barrel. Place a small disc of nylon gauze inside the barrel, to prevent the beads from blocking the syringe outlet.
- Separate the beads of immobilised enzyme from the liquid with the tea strainer.
- Carefully tip the beads into the syringe barrel.
- Close the tubing on the syringe barrel using a tubing clip.
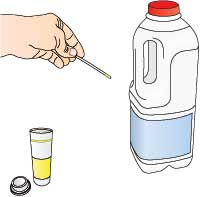
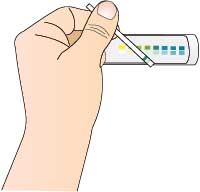
- Test the milk before treatment using the glucose test strips, to ensure that it does not contain any glucose.
- Pour a small volume of milk over the enzyme beads, then undo the clip and allow the treated milk to run into a small beaker.
- Test the milk leaving the column using the glucose test strips. If necessary, return the treated milk to the column until the desired concentration of glucose is achieved.

Safety guidelines
Do not consume the milk
The enzyme suggested for this work is safe to use, provided it is handled appropriately. Although Novozymes Lactozym® is a food-grade product, milk prepared using it should not be consumed. This is because the enzyme has not been handled aseptically, so it (and the product made using it) may have been contaminated.
Readers are advised to refer to any local safety guidelines and to carry out their own risk assessment for any practical work.
General enzyme safety guidelines

As enzymes are water-soluble, water should always be used for their removal if they are spilt.
Do not let liquid enzyme preparations dry up
If liquid preparations are allowed to dry up, there is a risk of dust formation. In susceptible people, the repeated inhalation of such dust may provoke asthma or a reaction similar to hay fever. Any spillage – on equipment, the floor or the bench – should be rinsed away immediately with water
Avoid the formation of aerosols
If enzyme-containing aerosols are formed, there is a risk of inhalation of the enzyme. In susceptible people, the repeated inhalation of such aerosols may provoke asthma or hay fever. For this reason, enzyme preparations should never be sprayed.
Avoid direct skin and eye contact
If, by accident, you get liquid enzyme on your skin or in your eyes, the remedy is plenty of tap water. The same applies to clothing. In the event of a spill on clothes, rinse with water then wash as usual. This treatment will generally prove sufficient, but if symptoms develop in the respiratory passages, on the skin or in the eyes, consult a doctor immediately.
Troubleshooting
Some UHT milk will test positive for glucose, probably because the heat treatment hydrolyses some of the lactose. UHT milk should therefore be avoided.
Additional investigations
The immobilised enzyme column may also be used to treat whey, producing a sweet whey syrup which is widely used in confectionery (it is usually described on labels as ‘hydrolysed whey syrup’ or just ‘whey syrup’).
Lactase is strongly inhibited by galactose (one of the products of its action on lactose). As a result, the flow rate of the substrate over the column is critical to the rate of the enzyme-catalysed reaction: too fast and there isn’t time for the reaction to occur; too slow and galactose will accumulate and then inhibit the reaction. Students can therefore investigate the effect of the flow rate on the conversion of lactose to glucose and galactose.
Suppliers
The NCBEw2 supplies Novozymes enzyme productsw3 to schools and colleges in the UK. Similar arrangements may exist in other countries.
Storage of materials
The enzyme preparations should be stored, undiluted, at 3-4 °C.
Web References
- w1 – A comprehensive description of lactose intolerance can be found on Wikipedia: http://en.wikipedia.org/wiki/Lactose_intolerance
- w2 – The National Centre for Biotechnology Education (NCBE) in the UK offers educational resources and practical training for teachers in several European Union countries. See: www.ncbe.reading.ac.uk
- w3 – For the website of Novozymes A/S, Denmark, see: www.novozymes.com
- w4 – This and other protocols are available for download from the Volvox website: www.eurovolvox.org
Resources
- Bayless TM, Paige DM, Ferry GD (1971) Lactose intolerance and milk drinking habits. Gastroenterology 60: 605–608.
- Richmond ML, Gray JI, Stine CM (1981) Beta-galactosidase: Review of recent research related to technological application, nutritional concerns and immobilization. Journal of Dairy Science 64: 1759–1771.
- Woodward J (ed; 1985) Immobilised enzymes and cells: a practical approach. Oxford, UK: Oxford University Press. ISBN: 0947946217. (An academic laboratory manual describing methods of immobilising enzymes and cells)
Review
This is one of a series of excellent biotechnology protocols from the NCBE, which bring the theory to life and illustrate many useful industrial applications of biotechnology at the laboratory scale and in short timeframes. The protocols are available to download from the Volvox websitew4.
Teachers will benefit greatly in their treatment of biotechnology from using this and similar protocols, and will be rewarded for the preparation required in stocking up on the necessary materials by the way in which the practical sessions bring the theory to reality. All of the protocols give satisfaction to the student investigators as they produce tangible results that are relevant to their everyday lives. Try it and see!
Marie Walsh, Republic of Ireland





