Diabetes mellitus Understand article
The incidence of diabetes is on the rise, in both the developed and developing worlds. Klaus Dugi, Professor of Medicine at the University of Heidelberg, Germany, discusses the causes, symptoms and treatment of diabetes.
Image courtesy of Dean
Madden
Translated from ancient Greek, diabetes mellitus means ‘honey sweet flow’ and stems from a time in which tasting a patient’s urine was still part of the physician’s diagnostic repertoire. By the sweet taste of the urine, diabetes mellitus could be distinguished from diabetes insipidus, another disease with increased urinary output.
The term diabetes mellitus covers several disease states with underlying causes that are characterised by an abnormal increase in blood sugar (glucose) levels.
Classification
In 1997, the World Health Organization and the American Diabetes Association agreed on a new classification for diabetes mellitus. The most frequent diabetes forms are Type 1 diabetes and Type 2 diabetes. Type 1 diabetes typically manifests during childhood or adolescence, but it has recently become apparent that adults can also develop this form, in some cases as late as in their forties or fifties. Type 2 diabetes is mainly a disease of the elderly, but as rates of obesity increase, more and more young adults, and even adolescents, are being diagnosed with this form of the disease. Other forms of diabetes include gestational diabetes (pregnancy diabetes), diabetes after operative resection of the pancreas, and rare forms of genetic diabetes.
Epidemiology
About 90% of patients with diabetes mellitus have Type 2 diabetes. An estimated 150 million people worldwide have Type 2 diabetes, and this number is expected to double within the next 20 years. Most of the increase will stem from developing and threshold countries such as India and China. In the USA, where the prevalence of diabetes is high, it is estimated that one in three people will develop Type 2 diabetes.
Pathophysiology
Normal situation
The body derives glucose from several sources. Firstly, it is taken up from food in the gut, either directly as glucose or after more complex carbohydrates have been digested into glucose and other simple sugars. Secondly, glucose is synthesised from other energy sources, mainly in the liver, in a process known as gluconeogenesis. Thirdly, glucose is stored in the liver, muscles, and other tissues in the form of glycogen. On demand, glycogen is broken down into glucose and secreted into the bloodstream.
Glucose is transported via the blood, whence it is absorbed by tissues needing glucose. After a meal, about 80% of the uptake of blood sugar is by muscle cells. During fasting, however, over 50% of glucose uptake is by the brain. This imbalance is probably because the sole source of energy for the brain is glucose , and the body needs to safeguard the brain’s glucose supply. If the blood sugar level falls too low (hypoglycaemia), the brain malfunctions, causing such symptoms as lack of concentration, dizziness and faintness, or, in severe cases, convulsions, coma and death.
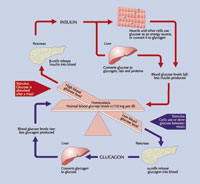
Image courtesy of Dean
Madden
The processes of glucose uptake and release are tightly regulated by hormones. The most important hormone is insulin, because it is the only hormone that can lower blood sugar levels. Insulin is synthesised and secreted only in the beta or islet cells of the pancreas. It lowers the blood glucose level by facilitating the uptake of glucose from blood by all cell types, reducing gluconeogenesis, and encouraging the storage of glucose in the form of glycogen.
Several other hormones, such as adrenaline, glucagon, thyroid hormone and growth hormone, are involved in increasing the blood sugar level.
Insulin resistance
The vast majority of patients with Type 2 diabetes or pre-diabetes are characterised by insulin resistance. Despite normal or even elevated blood levels of insulin, insulin has less effect than in normal individuals. Peripheral insulin resistance means that insulin is less effective at mediating the uptake of blood glucose by muscle cells. In the liver, insulin fails to suppress the production of new glucose and the breakdown of glycogen. Thus, insulin resistance tips the glucose homeostasis towards high blood sugar levels (hyperglycaemia).
Type 1 diabetes
Type 1 diabetes is an autoimmune disease. In genetically susceptible individuals, an inflammation of pancreatic beta cells is triggered, most likely by a viral infection. Because beta cells are the only ones able to produce and secrete insulin, complete insulin deficiency ensues. As a result, all Type 1 diabetic patients require insulin replacement therapy. The only exception is during the ‘honeymoon period’ that some patients experience shortly after diagnosis and initial treatment, owing to some residual insulin secretion early in the progress of the disease.
Type 2 diabetes
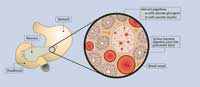
Image courtesy of Dean
Madden
Type 2 diabetes is a prime example of a disease caused by a combination of genetic and environmental factors. The genetic influence is greater than for Type 1 diabetes: the identical twin of a Type 2 diabetic patient is almost certain to develop the disease. On the other hand, lifestyle factors such as diet and exercise are also important determinants; in times of scarce food supply, for instance, the incidence of Type 2 diabetes is very low.
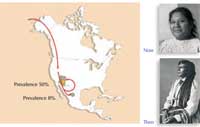
Pima Indians:
gene-environment
interaction
A good example of the interplay of genetics and lifestyle are the Pima Indians. Those living in Mexico have a diabetes prevalence of about 8%, whereas those who have emigrated to the USA, where the lifestyle is more sedentary and access to energy-dense (fatty) food is easier, have a diabetes prevalence of about 50%.
The most important risk factor for Type 2 diabetes is obesity. Epidemiological studies have shown that, compared to lean individuals, very obese men and women (body mass index >35) have a 60- and 90-fold increased probability of developing Type 2 diabetes, respectively (see figure). In terms of genetics, Type 2 diabetes is a multifactorial disease for which no single gene is responsible.

Type 2 diabetes: relative risk
for Type 2 diabetes (age
corrected)
In contrast to patients with overt Type 2 diabetes, patients with pre-diabetes (characterised by insulin resistance) do not exhibit hyperglycaemia when fasting. However, after a challenge like an oral glucose tolerance test (oGTT), during which 75 grams of glucose are ingested, patients exhibit pathologically high glucose levels (see Table 1). These patients, therefore, are characterised as having impaired glucose tolerance.
For a limited period, pancreatic beta cells are able to produce enough insulin to overcome the insulin resistance, so many pre-diabetic patients actually have elevated plasma insulin levels. However, in most patients, the rate of beta-cell death exceeds that of beta-cell formation in the pancreas, resulting in fewer insulin-producing beta cells. When the insulin-producing capacity of the pancreas is overtaken by the increased demand caused by insulin resistance, the patient develops overt Type 2 diabetes.
Three main factors contribute to hyperglycaemia:
- Insulin resistance in the muscle tissue, causing the muscles to take up less glucose from the blood.
- Impaired insulin secretion by the pancreas.
- Increased glucose production by the liver as a consequence of hepatic insulin resistance.
In the last few years, it has been demonstrated that adipose (fat) tissue and the central nervous system also play important roles in the pathogenesis of Type 2 diabetes.
Diagnosis
Symptoms
Unspecific early symptoms of diabetes include fatigue, a lack of general well-being, or an increased tendency towards infections, e.g. bladder infections. When hyperglycaemia becomes more pronounced, patients lose glucose via the urine, and produce more urine. This leads to the symptoms of overt diabetes, which are typically the initial symptoms of Type 1 diabetes: increased frequency of urination leading to increased thirst, and subsequently to dehydration and weight loss.
Screening
In the early stages of Type 2 diabetes, patients have few or no symptoms, and often go undiagnosed for many years. Unfortunately, diabetic complications (see below) frequently develop during this time. Therefore, it is important to screen those at risk of developing Type 2 diabetes, such as obese people, those with a family history of diabetes, and women who previously experienced gestational diabetes. Screening can be done either by measuring the blood glucose levels while the individual is fasting, or by performing an oGTT (see above). The most important criteria for diagnosing diabetes are shown in Table 1.
| Normal | Impaired fasting glucose or glucose tolerance | Overt diabetes | |
|---|---|---|---|
| Fasting glucose | <110 mg/dl <6.1 mmol/l |
110-125 mg/dl 6.1-6.9 mmol/l |
≥126 mg/dl ≥7.0 mmol/l |
| 2 h after oGTT | <140 mg/dl <7.8 mmol/l |
140-199 mg/dl 7.8-11.1 mmol/l |
≥200 mg/dl ≥11.1 mmol/l |
The most important measure for monitoring the course of diabetes is glycosylated haemoglobin (HbA1c). The higher the blood glucose value over time, the more non-enzymatic glycosylation of haemoglobin will occur. Because haemoglobin is transported in red blood cells, which have a mean lifespan of about 120 days, the HbA1c value is a reflection of the glucose control over the previous three months. In most assays, an HbA1c value of <6.1% is considered normal. The target value for diabetic patients is an HbA1c of <7.0%, or even <6.5%.
Treatment
The basis of diabetes treatment is educating patients about the pathogenesis of the disease, diabetic complications, dietary and drug treatment, and other aspects of the disease.
All diabetic patients need to be instructed about the appropriate diet and exercise, and the necessity, in most cases, of losing weight. Those Type 2 diabetic patients who manage to radically change their lifestyle and lose significant amounts of weight have a very good chance of ridding themselves of the disease. Unfortunately, only a very small minority of patients manage this.
If the HbA1c value stays above 7.0% despite improved diet and exercise, drug treatment is warranted. Several oral anti-diabetic drugs are currently on the market which target different causes of hyperglycaemia. Metformin reduces hepatic glucose output, sulfonylurea drugs increase pancreatic insulin secretion, and the glitazones reduce peripheral insulin resistance.
If the HbA1c value stays above 7.0% despite treatment with several oral anti-diabetic drugs, insulin therapy is indicated. The first step is often to treat with long-acting insulin at bedtime. Eventually, many Type 2 diabetic patients will, like Type 1 diabetic patients, require full insulin therapy. This will be either a fixed twice-daily regimen of mixed insulin containing a short-acting and a long-acting insulin, or an intensive regimen with injection of a long-acting insulin at bedtime and/or in the morning, and injections of short-acting insulin with meals. Insulin dose and food intake must be closely matched to prevent either hyper- or hypoglycaemia.
Due to the high risk of cardiovascular problems (see below), it is important to treat not only the glucose disturbances, but also other risk factors of cardiovascular disease such as high blood pressure and high cholesterol levels.
Complications
Due to a high frequency of complications, diabetes significantly reduces life expectancy. Because of complications with the small blood vessels (microvascular disease), Type 2 diabetes is currently the most frequent cause of adult-onset loss of vision (diabetic retinopathy), renal failure (diabetic nephropathy), and amputation (diabetic foot) in the industrialised world. The most frequent microvascular complication is diabetic neuropathy, usually a disease of the distal sensory nerves, which impairs the perception of vibration, temperature, and pain in feet and hands. In later stages, diabetic neuropathy can be characterised by severe pain.
In addition, Type 2 diabetes is associated with complications of the large blood vessels (macrovascular disease), and a two- to five-fold increased risk of cardiovascular disease, mainly myocardial infarctions (heart attack) and strokes.
Outlook
New application methods of insulin – inhaled and oral – are currently being tested in clinical trials, along with drugs with improved efficacy and reduced side-effect profiles.
More important, however, would be preventive measures to tackle the worldwide increase in obesity and diabetes, especially in developing countries in which most of the predicted new cases of diabetes will occur.
Review
This article is suitable for updating secondary school biology teachers on diabetes mellitus, an issue of general interest and social relevance. This disease is becoming more and more common in industrialised countries and even in developing countries.
The text, which is divided into paragraphs to make information retrieval easy, takes into account every aspect of the disease, is written in a plain style, and explains technical terms clearly.
I recommend the use of this material for the teacher as a basis for classroom activities on human physiology and health education. Upper secondary school students would also benefit from using the article as a source of information to work autonomously.
Giulia Realdon, Italy





